TORONTO and HAIFA, Israel, March 14, 2025 (GLOBE NEWSWIRE) — NurExone Biologic Inc. (TSXV: NRX) (OTCQB: NRXBF) (FSE: J90) (“NurExone” or the “Company”) is pleased to announce that it has successfully completed an important preclinical study towards its Investigational New Drug (“IND”) submission. The new study, which advances the Company’s path towards first-in-human trials, demonstrated that ExoPTEN treatment with different dose regimens led to both motor function recovery and significant improvements in blood flow at the site of spinal cord injury—an essential factor in tissue healing and functional recovery.i
“This preclinical study evaluated dosing regimens to provide efficacy data in support of our IND submission,” said Dr. Tali Kizhner, Director of R&D at NurExone. “The results reinforce ExoPTEN’s potential to enhance the body’s natural repair mechanisms following spinal cord injury. Notably, the increased blood vessel size observed in treated subjects indicated improved circulation, which is crucial for oxygen and nutrient delivery to damaged tissues. These findings suggest that ExoPTEN has the potential to become a transformative therapeutic candidate, and we are eager to advance toward clinical trials.”
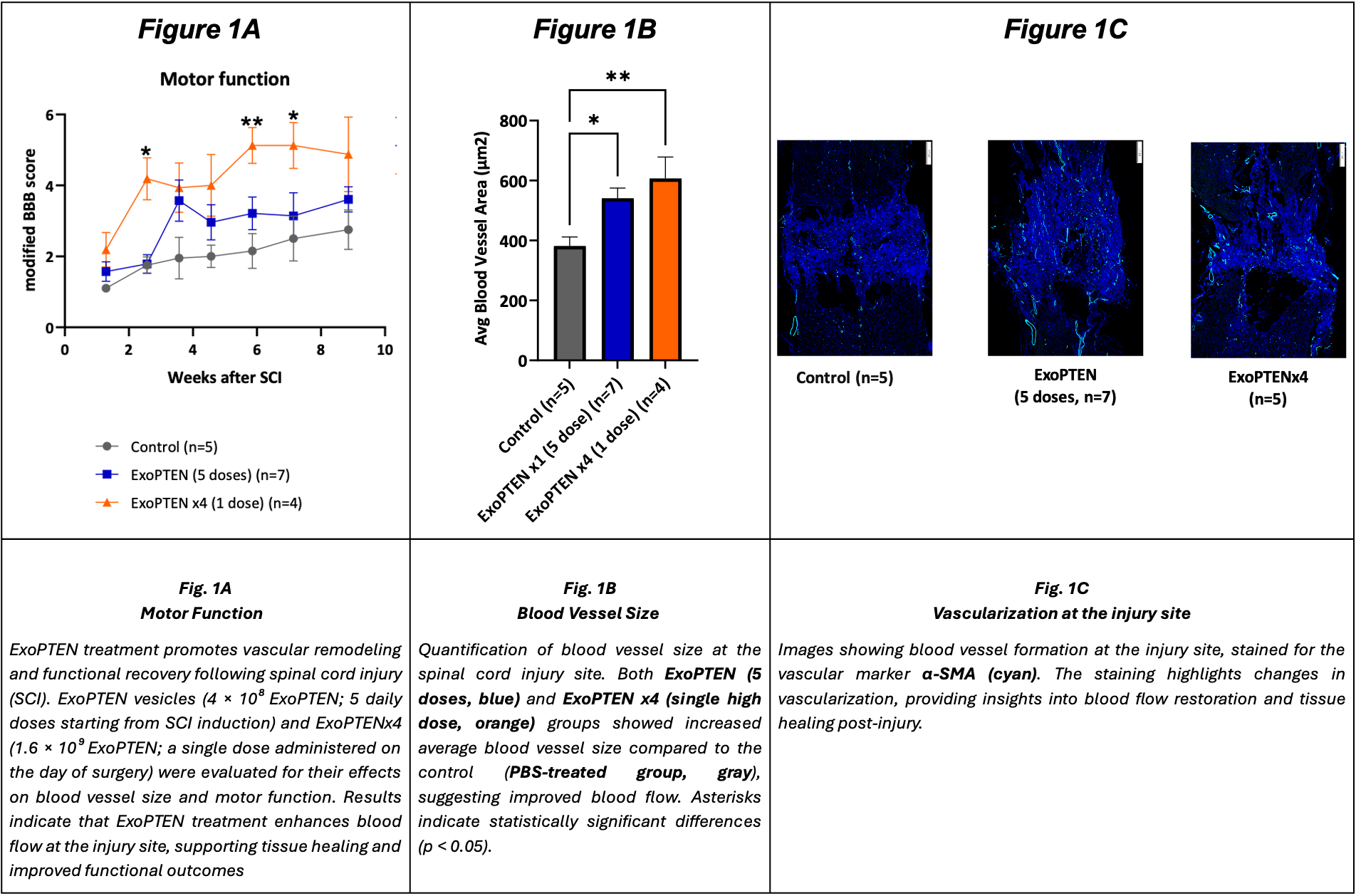
Scientific publications and reach in the field have shown already that post-injury angiogenesis and vascular remodeling correlate with improved functional recovery in spinal cord injury models.ii
The study compared two dosing regimens of ExoPTEN: a single high dose on the day of surgery versus a lower dose administered over five consecutive days. Both treatment groups showed significant improvements in motor function recovery compared to the control group, as measured by the modified Basso, Beattie, and Bresnahan (“BBB”) locomotor rating scale (Figure 1A). Additionally, histological analysis revealed that ExoPTEN treatment significantly increased the average blood vessel size (Figure 1B-1C), suggesting improved circulationi – a critical factor in post-injury healing and functional restoration.
NurExone will continue to refine ExoPTEN’s therapeutic profile as part of its ongoing preclinical program, paving the way to IND submission and regulatory approval for first-in-human trials.
About NurExone
NurExone Biologic Inc. is a TSX Venture Exchange (“TSXV”), OTCQB and Frankfurt-listed biotech company focused on developing regenerative exosome-based therapies for central nervous system injuries. Its lead product, ExoPTEN, has demonstrated strong preclinical data supporting clinical potential in treating acute spinal cord and optic nerve injury, both multi-billion-dollar marketsiii. Regulatory milestones, including Orphan Drug Designation, facilitate the roadmap towards clinical trials in the U.S. and Europe. Commercially, the Company is expected to offer solutions to companies interested in quality exosomes and minimally invasive targeted delivery systems for other indications. NurExone has established Exo-Top Inc., a U.S. subsidiary, to anchor its North American activity and growth strategy.
For additional information and a brief interview, please watch Who is NurExone?, visit www.nurexone.com or follow NurExone on LinkedIn, Twitter, Facebook, or YouTube.
For more information, please contact:
Dr. Lior Shaltiel
Chief Executive Officer and Director
Phone: +972-52-4803034
Email: [email protected]
Oak Hill Financial Inc.
2 Bloor Street, Suite 2900
Toronto, Ontario M4W 3E2
Investor Relations – Canada
Phone: +1-647-479-5803
Email: [email protected]
Dr. Eva Reuter
Investor Relations – Germany
Phone: +49-69-1532-5857
Email: [email protected]
Allele Capital Partners
Investor Relations – U.S.
Phone: +1 978-857-5075
Email: [email protected]
FORWARD-LOOKING STATEMENTS
This press release contains certain “forward-looking statements” that reflect the Company’s current expectations and projections about its future results. Wherever possible, words such as “may”, “will”, “should”, “could”, “expect”, “plan”, “intend”, “anticipate”, “believe”, “estimate”, “predict” or “potential” or the negative or other variations of these words, or similar words or phrases, have been used to identify these forward-looking statements. Forward-looking statements in this press release include, but are not limited to, statements relating to: the results of the Company’s preclinical trials; the Company completing an IND submission; the Company advancing towards clinical trials and launching a first-in-human trial; the anticipated benefits of ExoPTEN; the Company continuing to refine its product candidates; the Company receiving requisite regulatory approvals; the Company’s future plans and expectations; NurExone’s focus on developing regenerative exosome-based therapies for central nervous system injuries; and the NurExone platform technology offering solutions to companies interested in quality exosomes and minimally invasive targeted delivery systems for other indications.
These statements reflect management’s current beliefs and are based on information currently available to management as at the date hereof. In developing the forward-looking statements in this press release, we have applied several material assumptions, including: the Company will realize upon the stated benefits of the preclinical trials and such clinical trials will have the intended results; the Company will complete an IND submission; the Company will advance towards clinical trials and launch a first-in-human trial; ExoPTEN will have its anticipated benefits; the Company will continue to refine its product candidates; the Company will receive requisite regulatory approvals; the Company will continue to succeed; NurExone will continue to focus on developing regenerative exosome-based therapies for central nervous system injuries; and the NurExone platform technology will offer solutions to companies interested in quality exosomes and minimally invasive targeted delivery systems for other indications.
Forward-looking statements involve significant risk, uncertainties and assumptions. Many factors could cause actual results, performance or achievements to differ materially from the results discussed or implied in the forward-looking statements. These risks and uncertainties include, but are not limited to risks related to: the Company’s early stage of development; lack of revenues to date; government regulation; market acceptance for its products; rapid technological change; dependence on key personnel, and on the Company’s strategic partners; the fact that preclinical drug development is uncertain, and the drug product candidates of the Company may never advance to clinical trials or human trials; the fact that results of preclinical studies and early-stage clinical trials may not be predictive of the results of later stage clinical trials; the uncertain outcome, cost, and timing of product development activities, preclinical studies and clinical trials of the Company; the uncertain clinical development process, including the risk that clinical trials may not have an effective design or generate positive results; the inability to obtain or maintain regulatory approval of the drug product candidates of the Company; the introduction of competing drugs that are safer, more effective or less expensive than, or otherwise superior to, the drug product candidates of the Company; the initiation, conduct, and completion of preclinical studies and clinical trials may be delayed, adversely affected or impacted by unforeseen issues; the inability to obtain or maintain intellectual property protection for the drug product candidates of the Company; risks that the Company’s intellectual property and technology won’t have the intended impact on the Company and/or its business; the Company’s inability to carry out its preclinical trials and/or realize upon the stated benefits of the preclinical trials and/or such preclinical trials will not have the intended results; the inability of the Company to fulfill its intended future plans and expectations; the Company will be unable to complete an IND submission; ExoPTEN will not have its anticipated benefits; the Company will be unable to refine its product candidates; NurExone being unable to focus on developing regenerative exosome-based therapies for central nervous system injuries; the NurExone platform technology being unable to offer solutions to companies interested in quality exosomes and/or minimally invasive targeted delivery systems for other indications; and the risks discussed under the heading “Risk Factors” on pages 44 to 51 of the Company’s annual information form dated August 27, 2024, a copy of which is available under the Company’s SEDAR+ profile at www.sedarplus.ca. These factors should be considered carefully, and readers should not place undue reliance on the forward-looking statements. Although the forward-looking statements contained in this press release are based upon what management believes to be reasonable assumptions, the Company cannot assure readers that actual results will be consistent with these forward-looking statements. These forward-looking statements are made as of the date of this press release, and the Company assumes no obligation to update or revise them to reflect new events or circumstances, except as required by law.
Neither TSXV nor its Regulation Services Provider (as that term is defined in the policies of the TSXV) accepts responsibility for the adequacy or accuracy of this release.
_______________________
i Blood-spinal cord barrier after spinal cord injury: relation to revascularization and wound healing, J Neurosci Res, 2003, https://pubmed.ncbi.nlm.nih.gov/14515352/
ii Blood-Spinal Cord Barrier in Spinal Cord Injury: A Review, J Neurotrauma, 2021, https://pubmed.ncbi.nlm.nih.gov/33292072/
iii Spinal cord injury, Glaucoma
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/72e6aea2-8b07-47ab-a582-fbc21aa63c72





